The expectations for room differential pressures to maintain air quality in pharmaceutical facility design are consistent and well defined from a regulatory perspective. However, there is no common approach to the design, monitoring, or alarming of area differential pressures. This article explores differential pressure concerns in aseptic manufacturing, or cleanroom classes B, C, and D.
Nicholas Haycocks

Related Articles
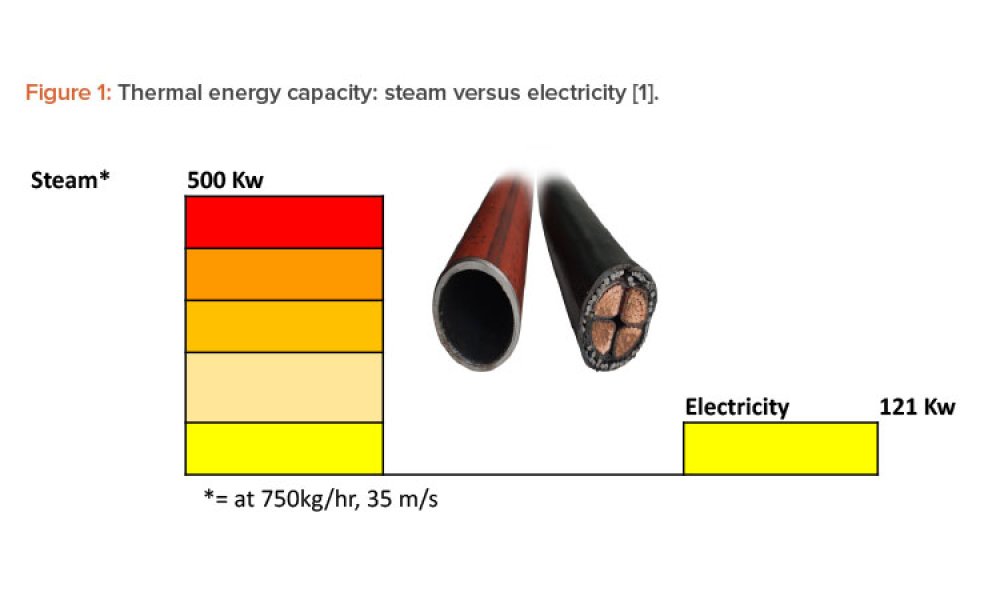
The world is beginning to grasp the huge challenge of achieving net-zero carbon emissions, or carbon neutrality, by 2050. Many countries have committed to achieving this ambitious goal. As a major global industry, the pharmaceutical sector has a significant role to play. For thermal energy–intensive industries, such as pharmaceutical manufacturing, the long-term future options to maintain...
Steam is the most powerful and effective thermal energy transfer fluid, and its use continues to grow in process industries around the world. However, there is very little written about the commissioning and qualification of pharmaceutical pure steam systems in GMP regulations or regulatory guidance. This article provides the background and science behind the steam quality tests and proposes a...
Defining room temperature and humidity limits is a frequent topic of debate when designing and operating pharmaceutical and biotechnology facilities. What are appropriate alarm limits and acceptable durations for an alarm condition? Understanding the source of temperature and humidity requirements, and strategies for setting limits, can ensure both compliance and optimum use of energy. This...


![Figure 1: Seasonal comfort (temperature and RH). ©ASHRAE, www.ashrae.org. Used with permission from 2017 ANSI/ASHRAE Standard 55 [4].](/sites/default/files/styles/teaser_image/public/2021-08/0921_PE_SO_TechHaycock_01.jpg?itok=mJiPGN_X)
