Quality Risk Management for Biopharmaceuticals

In the dynamic and highly regulated world of biopharmaceutical manufacturing, maintaining and ensuring quality is a critical success factor. An effective quality risk management (QRM) system is a key component in the overall quality management infrastructure of biopharmaceutical organizations. It offers a structured, scientific, and risk-based approach to decision-making, addressing potential quality issues during manufacturing. High performing organizations effectively implement QRM into overall quality policies and procedures to enhance and streamline decision-making.
Implementing a robust QRM system is more than just a compliance requirement. It fundamentally contributes to the organization’s commitment to patient safety, product quality, and data integrity. A robust QRM system consists of key characteristics with clearly defined processes that contribute to the system’s success.
Reviewing the Risk Compliance Data
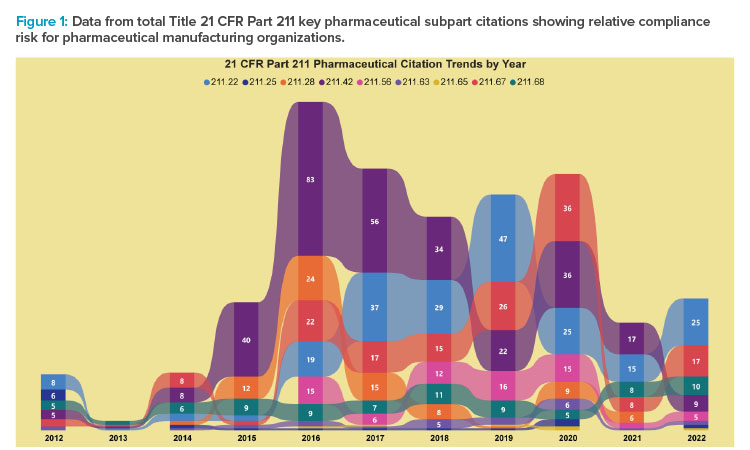
The following graphical data shows the relative compliance risk for pharmaceutical manufacturing organizations based on US Food and Drug Administration (FDA) regulatory activity (see Figure 1). Monitoring regulatory trends based on actual FDA activity provides useful insight for evaluating internal quality management system performance and proactively identifying areas of opportunity to improve overall compliance. Six major pharmaceutical regulation subparts are charted with relative annual activity increasing significantly from 2016 to 2020. During that period, the Building and Facilities, Laboratory Controls, and Production and Process Controls subparts were the largest areas, receiving 483 observations during regulatory inspections.
Those areas present increased compliance risk that would benefit from a formal review, gap analysis, and remediation to improve overall quality system performance and serve as priorities for time and resources. Regulatory risk during the COVID-19 pandemic decreased dramatically as the FDA performed few, if any, on-site investigations. However, activity during 2022 represents renewed on-site investigations with associated regulatory risk.
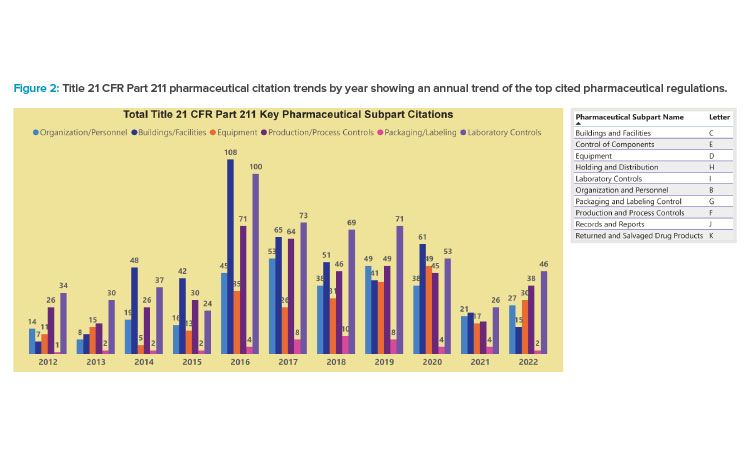
Figure 2 provides an annual trend of the top cited pharmaceutical regulations from 483 observations during regulatory investigations. These regulations are from the subparts identified in Figure 1 and the largest contributors to pharmaceutical regulatory risk. Any efforts to evaluate biopharmaceutical risk should consider the specific requirements identified in these regulations and address gaps identified during formal review and gap analysis as part of a QRM plan.
A Case Study
This case study concerns a major biopharmaceutical organization that specializes in producing monoclonal antibodies (mAbs) used in the treatment of various autoimmune diseases. As part of their commitment to quality and regulatory compliance, they have implemented a robust QRM system.
Challenge
The organization’s production engineers identified a potential risk in their manufacturing process. The risk was related to variability in the cell culture phase, which could potentially lead to inconsistencies in the final product’s efficacy and safety.
Risk Identification
The QRM team initiated the risk identification process using failure mode and effects analysis (FMEA) and brainstorming sessions with cross-functional teams. They identified key risk factors, such as pH imbalances, temperature fluctuations, and contamination risks during the cell culture phase.
Risk Assessment
Using a risk matrix, the team assessed the potential impact and likelihood of each identified risk. They determined that temperature fluctuations posed the highest risk due to their high likelihood and potential to significantly impact product quality.
Risk Control
The organization decided to implement additional control measures to mitigate this risk:
- Enhanced monitoring: Installing advanced temperature monitoring systems with automatic alerts for deviations.
- Process improvement: Optimizing the cell culture process to be more robust against minor temperature changes.
- Employee training: Conducting extensive training for staff on the importance of maintaining optimal temperature conditions.
Risk Communication
The QRM team communicated the identified risks, their potential impact, and the planned control measures to all relevant stakeholders, including the manufacturing team, quality assurance department, and senior management.
Implementation
The proposed measures were implemented, and their effectiveness was closely monitored. This included regular review meetings and updates to the risk management plan.
Outcome
The new control measures led to a significant reduction in temperature-related variability in the cell culture process. As a result, the consistency and quality of the mAbs improved, leading to enhanced patient safety and regulatory compliance.
Lessons Learned
The proactive approach to identifying and managing a critical risk in their manufacturing process demonstrated the importance of a dynamic and integrated QRM system. The case also highlighted the need for continuous monitoring and improvement in risk management practices.
Case Study Conclusion
This case study exemplifies the application of a structured QRM process in the biopharmaceutical industry. It illustrates the importance of identifying, assessing, controlling, and communicating risks in a systematic manner to ensure the production of high-quality biopharmaceutical products.
Characteristics of a Biopharmaceutical Qrm System
Identification of risk is a cross-functional effort that begins in the late development stages prior to technology transfer. In the early stages, research and development (R&D) is the main contributor in the risk identification process, which is facilitated by quality and manufacturing who are participants. As manufacturing develops detailed knowledge of the new process and technology, it provides a strong perspective on potential issues and risks that may exist in day-to-day manufacturing. At this time, all teams must compromise to ensure the final technology and process transfer meet the strategic goals of launching a new product.
Once the technology transfer is complete, manufacturing takes the lead in monitoring risk, along with quality. The manufacturing team also proposes any potential changes, which are reviewed by R&D, quality, and, possibly, commercial participants. Performance metrics are developed jointly between quality and manufacturing and used to periodically report to cross-functional leaders.
The main characteristics of a robust QRM system for biopharmaceutical manufacturers are identified in the following sections.
Risk Identification
The initial step in any QRM system is the identification of potential risks. It is necessary to understand what could potentially go wrong in the manufacturing process to manage and mitigate these risks effectively. Elevated performance in risk identification is demonstrated by organizations conducting risk identification with input from cross-functional subject matter experts.
This typically involves brainstorming sessions with relevant stakeholders, analysis of historical data and problem reports, and reviews of process documentation. Clear guidelines should be established for what constitutes a risk, and all identified risks should be documented and maintained in a risk register.
In addition to brainstorming sessions and historical data analysis, other tools such as FMEA, hazard identification, or process hazard analysis can be implemented for a systematic approach. Expert opinions and predictive models can also be used. A successful process should also involve reassessing the risk landscape periodically and after any significant changes. Changes requiring revalidation are a notable trigger to update risk profiles.
A robust biopharmaceutical QRM system recognizes that the process of risk identification is continuous and dynamic, adjusting to changes in procedures, equipment, materials, and the overall business environment. It also considers both internal and external sources of risk.
Risk Assessment
After identifying risks, it is crucial to evaluate them in terms of their potential impact on product quality and the probability of their occurrence. This allows the company to prioritize its risk management efforts.
Risk assessment usually involves qualitative or quantitative methods. Qualitative methods might include rating risks on a scale from low to high, whereas quantitative methods might involve statistical analysis or simulation. Risk assessment is about creating an informed understanding of the risk and considering the severity of the impact, the likelihood of occurrence, and the detectability of the risk. This aids in prioritizing resources and efforts for risk control.
The process should include risk ranking or scoring systems that can objectively evaluate and compare different risks. Detailed risk maps or matrices can be created to visualize the risk landscape. Risk assessments should be periodically reviewed and updated, especially when new information becomes available.
Risk Control
This step involves deciding on and implementing measures to mitigate the identified risks. Without this step, the risk management process would be incomplete. Risk control involves not only mitigating risks but also deciding whether to accept, transfer, or avoid certain risks. Risk control measures should be proportional to the significance of the risk.
Risk control could involve anything from making changes to the manufacturing process to training employees in new procedures. A key part of this step is documenting the control measures and monitoring their effectiveness over time. After devising risk control measures, a pilot test can be conducted for complex or high-stake measures to ensure their effectiveness before full-scale implementation. The measures should also be reviewed and updated regularly, and particularly after any significant incidents.
Communication and Consultation
Effective communication ensures all relevant stakeholders are aware of the risks and the steps being taken to control them. This not only fosters a culture of risk awareness, but also ensures risk management efforts are coordinated across the organization. Effective communication promotes a shared understanding of risks, risk management practices, and individual roles and responsibilities in managing risk. It should involve all levels of the organization, as well as external stakeholders when appropriate.
This could involve regular meetings, reports, or automated notifications. The key is to ensure that the right information reaches the right people at the right time. The communication process should be a two-way street, allowing feedback from all stakeholders. In addition to meetings and reports, knowledge management systems or collaboration platforms could be used to facilitate communication. Clear protocols should be established for escalation of high-priority risks.
Continuous Monitoring and Review
The risk landscape can change over time, with new risks emerging and old ones disappearing or changing in severity. Continuous monitoring and review ensure that a QRM system stays relevant and effective. Incorporating accurate trend data based on regulatory activity provides an additional level of input elevating the effectiveness of risk management activities.
This can involve regular risk assessments, audits, and reviews of risk control measures. Any changes should be documented and communicated to relevant stakeholders. Monitoring and review processes should include the risks themselves and the effectiveness of the QRM system, changes in context, and the identification of emerging risks.
Risk Management Integration
Risk management should be an integral part of all organizational processes—not a separate activity. This ensures risk considerations are a part of all decisions, rather than being an afterthought. Integrating risk management with other business processes ensures risk management is proactive rather than reactive. It allows risks to be addressed before they can cause problems.
This could involve incorporating risk management into existing process documentation, training employees on risk management, or establishing a risk management committee. This could involve the use of integrated management systems or embedding risk management into standard operating procedures. Cross-functional teams or committees could be established to oversee the integration. Key performance indicators related to risk management should be established and monitored. Audits and reviews should be scheduled regularly and triggered by significant changes or incidents. Feedback from these activities should be used to drive continuous improvement.
Root Cause Analysis
Understanding the root cause of a problem allows for more effective risk management. It helps avoid merely treating the symptoms of a problem, which can lead to recurrence. The goal of root cause analysis is to prevent recurrence of problems by addressing their underlying causes, not just the symptoms. It allows for more efficient use of resources and improves process understanding.
Techniques such as the five whys and fishbone diagrams, among others, can be used to identify root causes. Once identified, these root causes should be addressed in the risk control measures. When conducting root cause analysis, it is important to ensure a blame-free environment where all ideas are considered. Tools such as Pareto charts could be used to prioritize root causes. Root cause prioritization may also reference regulatory trends based on current regulatory activity. Corrective and preventive actions should be devised to address the root causes.
Data-Driven Decision-Making
Decisions about risk management should be based on data, not on gut feelings or intuition. This leads to more objective and effective decisions. The use of data promotes objectivity, consistency, and efficiency in decision-making. It also allows for tracking and demonstrating the performance of the QRM system. An example of data-driven decision-making used by high-performing organizations uses available newsletters, visualizations, and trend tracking regulatory data to provide accurate insights to compliance risk.
This might involve collecting and analyzing data on process performance, product quality, and the effectiveness of risk control measures. Decision-making tools such as decision trees or Bayesian networks can also be used. An effective process should include not only collection and analysis of data, but also data management practices to ensure data integrity and usability. Advanced data analytics or artificial intelligence could be used for predictive risk modeling. Regulatory trends and current regulatory activity are also indicators providing insight into predictive risks of regulatory audits.
Quality Culture
A strong culture of quality fosters individual accountability, intrinsic motivation, and proactive behavior in managing risk and it ensures risk management is not the responsibility of just the quality department. Successful organizations building a strong culture of quality and compliance have notable focus and support from executive leadership. A successful quality culture can only succeed with outstanding support from organizational executives. The “tone at the top” significantly drives the performance and adherence of the organization to quality principles.
This could involve training, recognition programs, or changes to organizational structure. It is important to regularly assess the culture of quality and adjust as needed. Activities to foster a quality culture could include workshops, training sessions, recognition programs, and team-building activities. Regular culture assessments could be conducted through surveys or interviews and the findings used to inform culture improvement initiatives.
A robust biopharmaceutical QRM system recognizes that the process of risk identification is continuous and dynamic, adjusting to changes in procedures, equipment, materials, and the overall business environment.
Flexibility and Adaptability
As the organization and its external environment change, the QRM system needs to be able to adapt. A rigid system that cannot handle change will quickly become ineffective. A flexible and adaptable QRM system allows the organization to respond effectively to changes and challenges, turning them into opportunities rather than threats. It helps ensure the system’s resilience and long-term sustainability. Using data-driven metrics and tracking tools facilitates effective management of quality and compliance risk.
This can involve regular reviews of the QRM system and a process for making changes to it. Feedback from stakeholders should be actively sought and incorporated. Scenario analysis or stress testing could be used to evaluate and improve the system’s adaptability. A change management process should be established to handle changes in a systematic and controlled manner. Integral to a change management process should be the incorporation of risk assessment and evaluation relevant to any proposed changes.
Compliance with Regulations
Biopharmaceutical companies operate in a heavily regulated environment. Compliance with regulations avoids legal problems and ensures products are safe and effective. It also promotes trust and credibility among stakeholders, and it provides a baseline for risk management practices.
Compliance can be ensured by keeping up to date with regulatory changes, incorporating these changes into the QRM system, and regularly auditing for compliance. Regular training should be provided to keep staff current on regulatory requirements. Regulatory intelligence activities could be conducted to anticipate and prepare for upcoming changes.
Compliance checks should be integrated into the risk assessment and review processes. Additionally, compliance reports and newsletters summarizing regulatory activity provide valuable insight into risks and trends associated with regulatory compliance for life sciences. Further expanding compliance data to broader time horizons increases insights into longer-term trends and the value of current trends in a historical perspective.
Traceability and Documentation
Documentation provides evidence of the QRM system’s functioning. It also allows for traceability, which is crucial for root cause analysis and for demonstrating compliance with regulations. Proper documentation allows the team to preserve institutional knowledge, learn from past experiences, and demonstrate compliance. Traceability is crucial for investigating incidents, validating processes, and ensuring product quality.
Documentation should be maintained for all risk management activities, including risk identification, assessment, and control. It should be kept in a format that is easily accessible and understandable. Traceability can be maintained through unique identifiers for risks and control measures, and by linking related documents. A document management system could be used to manage and control documents. The system should support version control, approval processes, and easy retrieval of documents. Traceability could be maintained through traceability matrices or dedicated software systems.
Conclusion
An effective QRM system in biopharmaceutical manufacturing is multifaceted, involving the identification and assessment of potential risks, robust control mechanisms, effective communication strategies, and regular monitoring and review procedures. The QRM system should be flexible and adaptable, grounded in data-driven decision-making, and deeply integrated within the organization’s culture and processes. The risk management process, which includes risk identification and mitigation, is a cross-functional effort requiring participation from R&D, quality, and manufacturing, with metrics for monitoring and reporting process effectiveness to cross-functional leaders.
Organizations performing at elevated levels consistently demonstrate an ability to incorporate risk criteria into daily operations using various tools to evaluate risk according to product and patient impact. Compliance with regulations and maintaining detailed traceability and documentation are also of paramount importance. Although implementing such a comprehensive system can be complex, the benefits of ensuring product quality and safety, and ultimately patient health, are profound. The successful deployment of QRM necessitates a continual commitment to each of these characteristics, fostering a culture of quality that permeates every aspect of the organization.


