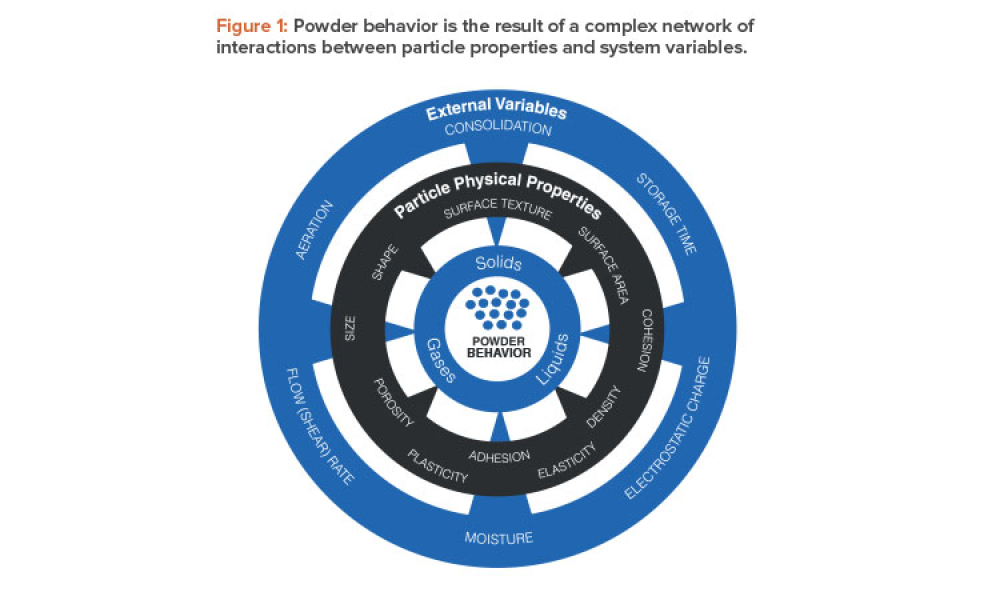
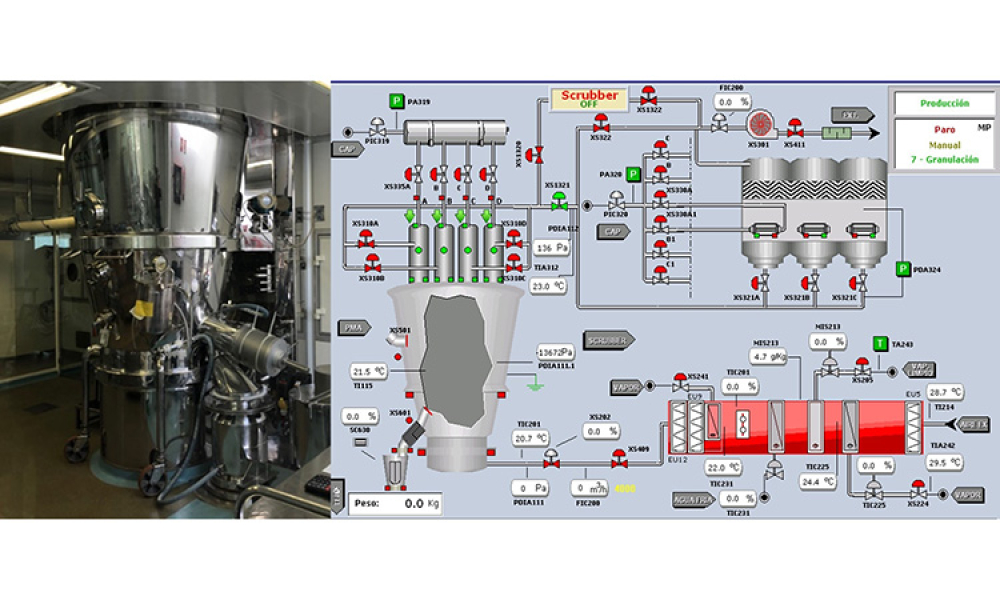
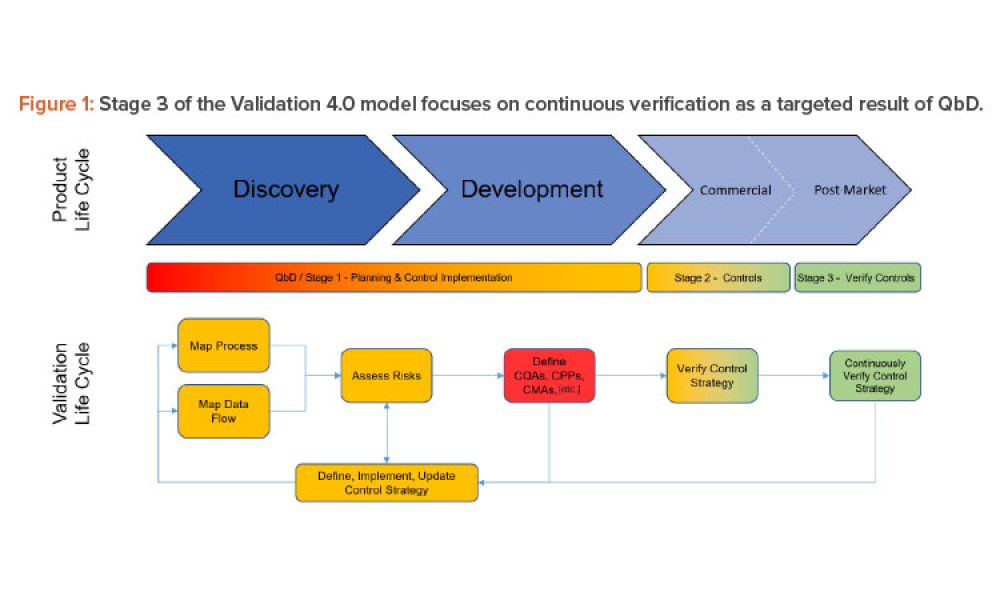
Oral Solid Dosage (OSD) is the inspection and evaluation of the manufacturing and control processes used to manufacture solid oral dosage form pharmaceutical products.
Guidance Documents
Advanced Manufacturing (1)
+Manufacturing Operations (2)
+Microbiological & Viral Contamination Control (1)
+Oral Solid Dosage (2)
+Community Discussions
Community Discussions
Mar 28, 2025
Information Systems
Regulatory
Advanced Manufacturing
Active Pharmaceutical Ingredients
Feb 13, 2025
Manufacturing Operations
Oral Solid Dosage
Nov 04, 2024
Regulatory
Supply Chain
Active Pharmaceutical Ingredients
Good Manufacturing Practice
Oct 31, 2024
Regulatory
Quality
Good Manufacturing Practice
Sustainable Facilities, HVAC, & Controlled Environments
Oct 23, 2024
Quality
Active Pharmaceutical Ingredients
Good Manufacturing Practice
Pharmaceutical Engineering Magazine Articles
White Papers
July / August 2024
The explosive growth of advanced therapy medicinal products (ATMPs), particularly cellular…
May / June 2024
The commercialization of personalized medicine has ushered in demand for a new type of facility—…
March / April 2024
Navigating the Asia Pacific Pharmaceutical Landscape for Global Impact Cover: The Asia Pacific…
January / February 2024
Stakeholders across industries are becoming accustomed to using information technology (IT) systems…
November / December 2023
There is much that large-scale commercial stem cell therapy processes can adopt from the existing…

iSpeak Blog Posts
Videos
Professional Development Training
ICH Q6A: Specifications, Test Procedures, and Acceptance Criteria for New Drug Substances and Products
+GMP Sterile Pharmaceutical Manufacturing Facility Training Course
+White Papers
July / August 2024
The explosive growth of advanced therapy medicinal products (ATMPs), particularly cellular…
May / June 2024
The commercialization of personalized medicine has ushered in demand for a new type of facility—…
March / April 2024
Navigating the Asia Pacific Pharmaceutical Landscape for Global Impact Cover: The Asia Pacific…