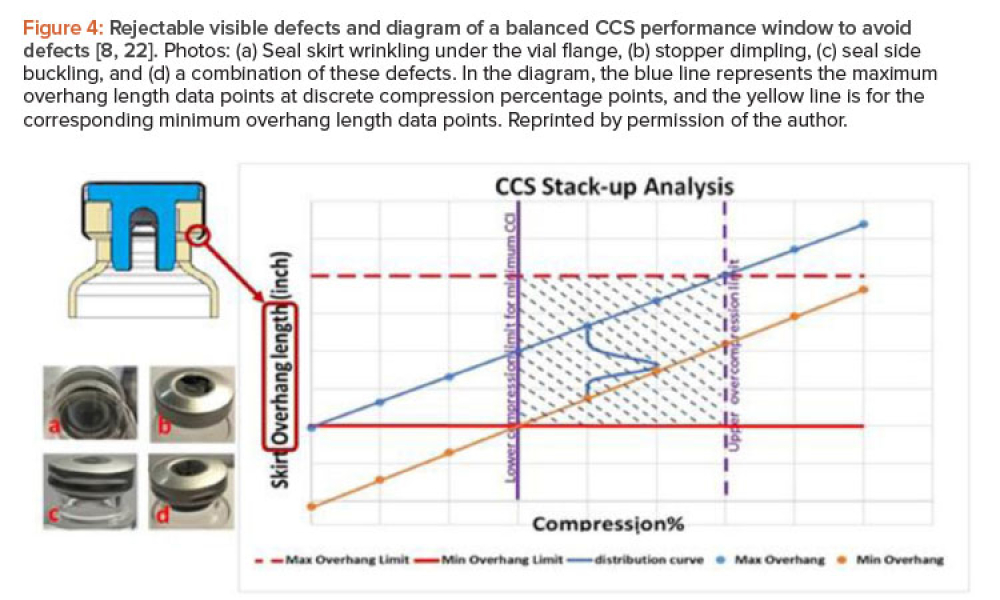
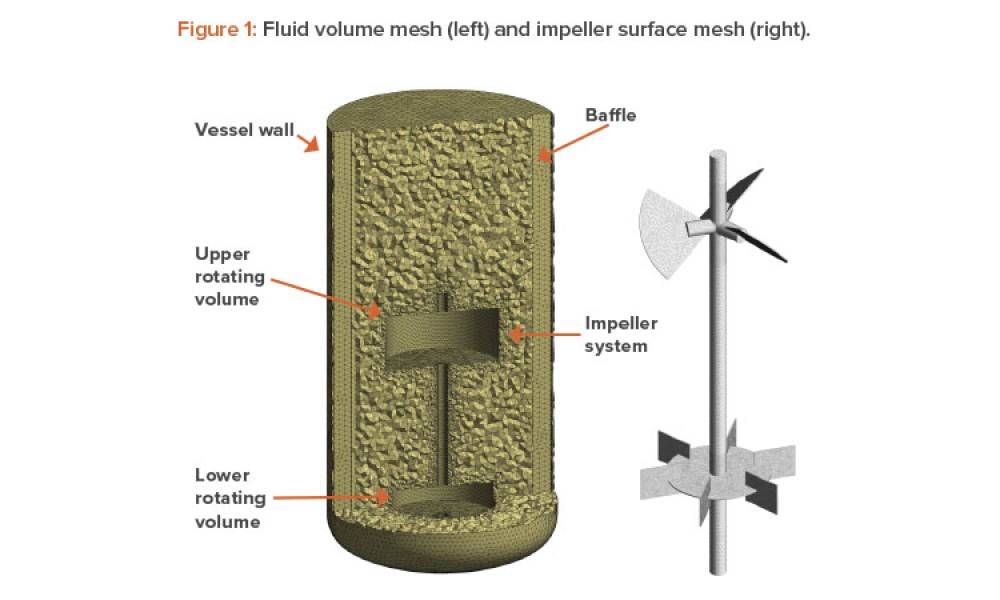
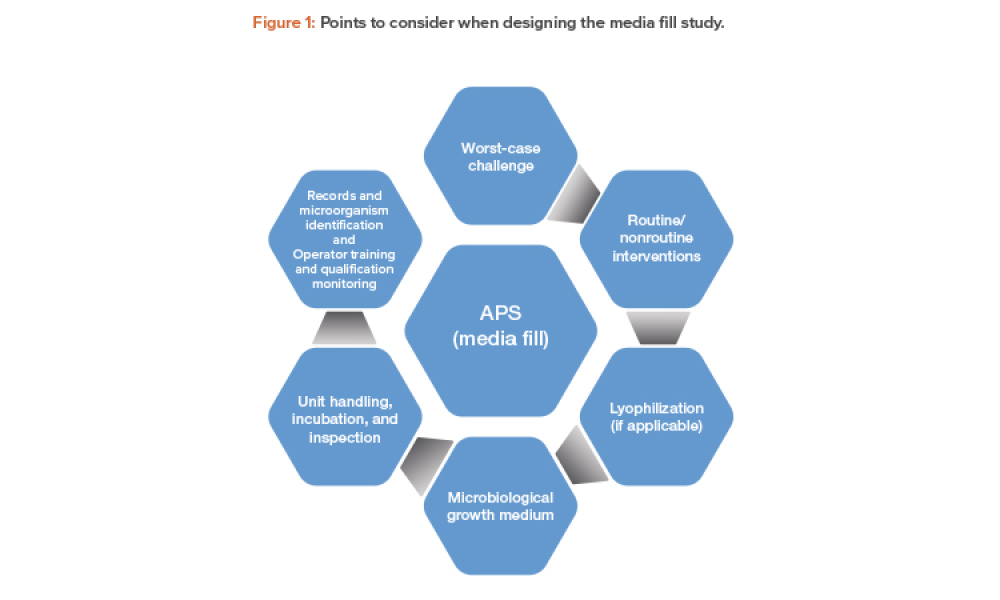
Sterile products processing relates to how sterile drug products are manufactured using aseptic (or free from contamination) process methods where the drug substance, excipients, and vehicle (e.g., saline or water for injection) are combined and filled into a container (such as a syringe)
Guidance Documents
Manufacturing Operations (1)
+Microbiological & Viral Contamination Control (1)
+Packaging (1)
+Sterile Products (2)
+Community Discussions
Community Discussions
Apr 16, 2025
Information Systems
Regulatory
Advanced Manufacturing
Artificial Intelligence
Apr 08, 2025
Validation
Apr 08, 2025
Data Integrity
Apr 07, 2025
Advanced Manufacturing
Artificial Intelligence
Mar 28, 2025
Information Systems
Regulatory
Advanced Manufacturing
Artificial Intelligence
Mar 20, 2025
Sustainable Facilities, HVAC, & Controlled Environments
Mar 20, 2025
Webinars
Upcoming
On-Demand
iSpeak Blog Posts
Pharmaceutical Engineering® Magazine
Videos
Professional Development Training
Aseptic Processing & Annex 1 Training Course
+White Papers
March / April 2025
A Skill Management Framework for a Pharma 4.0™ Workforce Feature: Pharma 4.0™ is driving fundamental…
January / February 2025
GAMP® is indispensable for safeguarding the safety, quality, and compliance of pharmaceutical…
November / December 2024
Plastic Process Waste in Biopharmaceutical Manufacturing Cover: This article presents a…